TRIS(2,4-DI-TERT-BUTYLPHENYL) PHOSPHITE
PRODUCT IDENTIFICATION
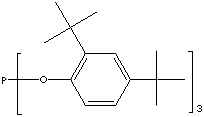
CLASSIFICATION
Polymer stabilizer, Plasticizer
EXTRA NOTES
Other RN:69344-92-9; 104381-89-7; 129038-69-3; 219315-40-9; 245439-51-4; 478284-78-5; 754233-11-9; 874911-33-8
PHYSICAL AND CHEMICAL PROPERTIES
183 - 187 C
REFRACTIVE INDEX
EXTERNAL LINKS & GENERAL DESCRIPTION
USA.gov - Tris(2,4-di-tert-butylphenyl) phosphite
Google Scholar Search - Tris(2,4-di-tert-butylphenyl) phosphite
PubChem Compound Summary - Tris(2,4-di-tert-butylphenyl) phosphite
KEGG (Kyoto Encyclopedia of Genes and Genomes) - Tris(2,4-di-tert-butylphenyl) phosphite
ChEBI (http://www.ebi.ac.uk/chebi/) - Tris(2,4-di-tert-butylphenyl) phosphite
NCBI (http://www.ncbi.nlm.nih.gov/) - Tris(2,4-di-tert-butylphenyl) phosphite
Material Safety Data Sheet - Tris(2,4-di-tert-butylphenyl) phosphite
EPA - Substance Registry Services - Tris(2,4-di-tert-butylphenyl) phosphite
Local:
Phosphorous acid is a clear to yellowish crystalline solid with a garlic like odour melting at 73 C, decomposes at 200 C. It is very soluble in water and in alcohol. This compound contains one direct P-H bond (which is not very acidic) and only two hydrogens bonded to oxygen (which are acidic). The structure of this material is more correctly written (HO)2HPO. For this reason, this dibasic acid forms two series of salts, one containing the dihydrogen phosphite ion, H2PO3- , and the other containing the hydrogen phosphite ion, HPO32-. It is prepared by hydrolysis of phosphorus trichloride (or tetraphosphorus hexaoxide) with alcohols or phenols. Phosphorous acid esters are called phosphite with the formula (RO)3P. Phosphorous acid and phosphite are used as reducing agents in chemical industry because of easy oxidation property to phosphoric acid. They are used as antioxidant, stabilizer and chelating agent in plastic system. They are used as solvent in paint and as flame retardant on fibres. They are used as a chemical intermediate in the production of pharmaceutical ingredients, pesticides, optical brighteners and in lubricant additives and adhesives. Tris(2,4-di-tert-butylphenyl) phosphite is used as a phosphite processing stabilizer for polycarbonate, polyolefins. It is used in combination with phenolic antioxidants acts for synergistical color stability and polymer viscosity. Antioxidant is a substance added in small quantities to hydrocarbons which are
susceptible to oxidation, such as rubbers, plastics, foods, and oils to inhibit or slow oxidative processes, while being itself oxidized. Antioxidants work in two different ways. In primary antioxidants (also called free-radical scavengers), antioxidative activity is implemented by the donation of an electron or hydrogen atom to a radical derivative. These antioxidants are usually hindered amines (p-Phenylene diamine, trimethyl dihydroquinolines, alkylated diphenyl amines) or substituted phenolic compounds with one or more bulky functional groups such as a tertiary butyl at 2,6 position commonly. Butylated hydroxytoluene (BHT) is a common example of hindered phenolic antioxidant. The reaction rate, or carbocation stability, in SN1 mechanism is 3° > 2° > 1° > CH3 (no SN1) so, tertiary alkyl moiety exists in lots of phenolic antioxidant compounds. Primary antioxidants are free radical scavengers which combine with peroxy radicals and break autocatalytic cycle. In secondary antioxidants ( also called peroxide decomposers), activity is implemented by the removal of an oxidative catalyst and the consequent prevention of the initiation of oxidation. Examples of peroxide decomposer type of antioxidant are trivalent phosphorous and divalent sulfurcontaining compound such as sulfides, thiodipropionates and organophosphites. Synergistic effect is expected when primary antioxidants are used together with secondary antioxidants as primary antioxidants are not very effective against the degradation by UV oxidation. Sometimes, chelating agents are added to scavenge metal impurities which can initiate decomposition.
Polymer stabilizer for polymers. Many polymers are susceptible to environmental degradation and require the addition of a stabilizer such as an antioxidant or UV absorber to prevent premature degradation. Hindered phenols and hindered amine light stabilizers (HALS) deactivate existing radicals, including peroxy compounds formed by air oxidation. UV absorbers dissipate UV radiation by a process nondestructive to the polymer. Free radical inhibitors are added to monomers as stabilizers to inhibit premature polymerization.
APPEARANCE
PURITY
99.0% min
MELTING POINT
181 C min
ACID NUMBER
HEAT LOSS
0.1% max
Not regulated
HAZARD OVERVIEW
GHS (Globally Harmonised System) Classification: Not a dangerous substance. Potential Health Effects: Eyes - May cause eye irritation. Skin - May be harmful if absorbed through skin May cause skin irritation. Inhalation - May be harmful if inhaled. May cause respiratory tract irritation. Ingestion - May be harmful if swallowed.
RISK PHRASES
36/37/38
SAFETY PHRASES
26-36